Passenger cars
Putting hybrid fluids to the test
21 April 2026
13 July 2016
New insights reveal the limitations of bench oil oxidation tests

Engine oil oxidation is a major oil degradation mechanism, which means it has been incorporated into both OEM and industry lubricant specifications. However, as Infineum's Dave Coultas explained at this year’s UNITI conference, the bench oxidation tests currently in use are unable to mimic the mechanisms of real engines and may restrict future formulation developments.
Oxidation is an inevitable consequence of the exposure of lubricants to high temperatures and pressures in highly reactive environments, such as those found in the piston zone and sump of modern internal combustion engines. In his 2016 UNITI presentation, Dave built upon the presentation he delivered in 2015, in which he examined the effect of oil degradation on lubricant performance [Read the Insight article here].
Since oxidation is one of the key oil degradation mechanisms, Infineum wanted to better understand if currently used bench oxidation tests produce realistic data. In addition, the work assesses whether the engine oils, which currently pass the laboratory tests, can protect the engine from oxidation under real conditions.
Both engine and bench tests have been developed to determine the effects of lubricant oxidation. Chassis dynamometer tests can stress the oil in a number of ways and use a variety of operating conditions, including different fuel type and composition, engine design and duty cycle, to mimic real-world conditions. However, these tests are expensive to run; typically in excess of $50,000 and, in many cases, tests need to run under extreme conditions in order to replicate field effects in a short period of time.
This led industry bodies, such as the CEC, and individual OEMs to develop a variety of oxidation bench tests, which have now been incorporated into both OEM and industry lubricant specifications. The introduction has been well intended with the key benefits of reduced test costs and less time required for lubricant performance evaluation. However, there are currently more than a dozen subtly different laboratory oxidation tests all with one common aim: to assess the oxidative resistance of lubricating oil in a standardised laboratory test.
The mechanisms of real engine oil oxidation are fundamentally different to those of bench oxidation tests.
During the wear process in a real engine for example, tiny wear iron particles are created. These fine particles react with combustion acids to form catalytically active Fe(III) species, which can accelerate oxidation. However, lubricants can be formulated to control the conversion of wear iron to active iron, thus limiting oxidation.
Bench oxidation tests have a difficult time replicating field oxidation mechanisms.
A key factor here is that the active Fe(III), which is in the form of Fe(AcAc)3, is added early on in the test, rather than over time as a result of wear. In addition these tests can be run at unrealistically high temperatures, between 150-170oC, and incorporate a wide range of conditions, durations and fuel types.
To better understand the value of the results produced in a laboratory, Infineum compared the oxidation mechanism and performance of lubricants in both real engines and bench oxidation tests.
For the comparison, oils were tested in both a chassis dynamometer test and the CEC L-109, a severe bench oxidation test, which will be used in ACEA specifications.
In an attempt to get closer to the conditions of the CEC L-109 test, the chassis dynamometer was set up with very harsh operating conditions. It replicated running a diesel van in the Infineum ‘Mountain Drive Cycle’, which simulates constant uphill driving on the Großglockner High Alpine Road in Austria, ascending to 2,504m. The test was run with high fatty acid methyl ester (FAME) sump doping and, over the 30,000 km, the cooling circuit was bypassed and no oil top up was carried out.
Despite this severe operation, it was not possible to mimic the bench test conditions in the engine, even with a prolonged test cycle.
For example, the average oil temperature in the engine test was 123oC peaking at 140oC, which is well below the 150-170oC set by the laboratory tests.
Two oils were formulated for testing: oil A controls the conversion of wear iron to active iron and oil B does not. Oil B was run in the chassis dynamometer until the oxidation increase reached the same level as oil B in the CEC L-109 oxidation test. When oil A was run for the same duration, CEC L-109 oxidation increase was 40% higher than in the chassis dynamometer test. This difference is a result of Fe(AcAc)3, a form of active iron added at the start of the CEC L-109 test, which removes the lubricant’s ability to control the conversion of wear iron to active iron.
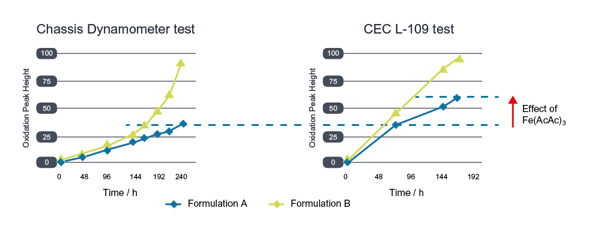
The effect of active iron on oxidation test results can be clearly seen
Looking at the viscosity increase response, a much bigger difference can be observed, with the CEC L-109 test generating a large viscosity increase when compared with the chassis dynamometer. This is down to the mechanism of viscosity increase, which in the bench test is disproportionally affected by the high temperatures and Fe(AcAc)3 present compared with the chassis dynamometer.
These results lead us to the conclusion that the CEC L-109 bench test, and the many other similar bench tests, although well intended, have limitations. By oversimplifying the variety of complex reactions taking place in a real engine, the ability to predict real engine performance can be lost.
Currently, the main drivers for engine oil developments are improving fuel economy, engine wear protection, cost reduction and more specific issues including low speed pre-ignition. While these drivers push oil formulations in one direction, unfortunately bench tests, like the CEC L-109, are driving the formulation space in exactly the opposite direction and can hinder innovation.
Bench oxidation tests might end up defining the formulation space based on one part of the oxidation mechanism. However, this does not necessarily mean the formulation approach used is the best way to improve the consequences of oxidation in a real engine environment.
In addition, these tests have a number of impacts on the lubricant formulator including:
The proliferation of bench oxidation tests creates unnecessary complexity and actively works against engine oil innovation to meet real industry needs. A key concern is that when the severity for bench tests is set too high, the main objective for oil formulation is to pass the non-predictive bench test. This could mean, for example, that too much additive is used or that the wrong type of additive solution is applied for truly relevant performance for an engine in the field.
It is clear that demonstrating field performance is now essential and, if bench tests remain at all, then realistic limits must be set that are also commensurate with the accuracy of the measurement tool.
Perhaps the time has come for industry to apply a high level of scrutiny and to conduct a cost/benefit analysis of bench and analytical tests to ensure they can model real field phenomena and are relevant to modern engines.
The results obtained in this study highlight the need for lubricants designed to meet future hardware requirements to provide excellent oxidation control and wear protection via the use of traditional additive technology. In addition, by better understanding the underlying mechanisms involved, more innovative approaches can be used to tackle these problems; such as the example here of controlling the conversion of wear iron to active iron. To achieve this balance, oil formulators need the freedom to select components and treat rates that can deliver optimum lubricant performance.
In our view, a holistic review of specifications is warranted to achieve a better balance of performance tests and to truly depict the necessary performance of lubricants.
Sign up to receive monthly updates via email
